The simplified notation led the way for English analytical chemist John Newlands to formulate his Law of Octaves and a prototype periodic table of the elements in 1864, but it was Russian chemist Dmitri Mendeleev who really laid it all on the table with 63 elements in 1869. So it's Au for gold and Ag for silver, not the circled G and S of Dalton's original notation. In an era when all Europe's learned men (and the few women who were allowed into schools and universities) knew Latin, the shared language was an international lingua franca.Īll but a handful of Berzelius' symbols are still used today. Berzelius organized 47 elements with letters alone, and he based those letters not primarily on the English names, but on the Latin ones. Half of Dalton's symbols used letters inside a circle to represent the element. One of the events that contradict the view that atoms cannot be fragmented and rebuilt is that atoms radiate alpha particles.A decade after Dalton formulated his symbols, Swedish chemist Jöns Jakob Berzelius simplified the system. Ītoms can break down as a result of nuclear reactions.Just as not all the atoms of an element are the same, not all the compound molecules are the same. The smallest known particle is not an atom. Not all the atoms of an element are the same since an atom can have several isotopes. What shortcomings and errors did Dalton's atomic theory have? If we compare 2g of hydrogen and 1g of hydrogen combined with the same amount of oxygen in both compounds, we get the number 2. For example: in H2O, while 2g of hydrogen atoms and 16g of oxygen atoms are combined, in OH, 1g of hydrogen and 16g of oxygen are combined. The Dalton’s law of multiple proportions: If two elements make up more than one compound, there is a simple relationship between the different amounts of the two elements combined, which can be expressed as whole numbers by weight. The sum of the atomic weights remains constant. Law of conservation of mass: The sum of the masses of the substances that react in a chemical reaction is equal to the sum of the masses of the substances (products) released. What are the laws related to Dalton's atomic model?ĭalton's atomic theory also allows a better description of the problems of chemical change: Ĭhemical reactions consist solely of the separation, union, or rearrangement of atoms.Ītoms of different elements can combine to form other chemical compounds.The atoms of different elements vary in size and atomic mass. Īll the atoms of a particular element are identical with the same atomic mass.What is John Dalton's atomic theory based on?ĭalton's atomic theory is based on four assumptions Ĭhemical elements are made up of tiny, indivisible, and indestructible particles called atoms. 
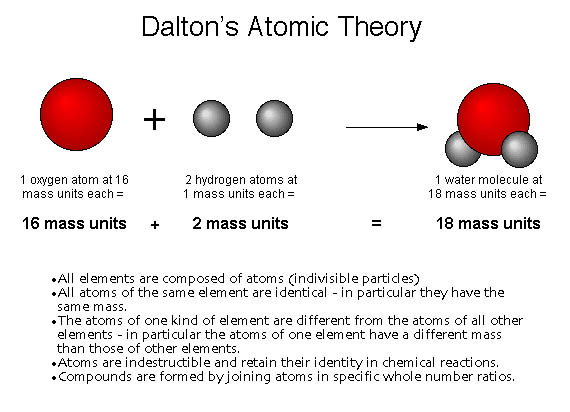
Modern atomic theory is based on Dalton's theory, but parts of it have been changed. His book “A New System of Chemical Philosophy” was the first application of atomic theory to chemistry. However, this time it was based on a series of scientific laboratory experiences.ĭalton was a member of the Manchester Literary and Philosophical Society since 1793. In 1808, John Dalton developed and published his theory of the atom, which took up the old ideas of Leucippus and Democritus. Dalton also found the relative masses of the atoms. According to this atomic theory, compounds are formed due to the combination of two or more elements in a simple proportion. The atoms of different chemical elements are different from each other. According to Dalton's atomic theory, the matter is made up of indivisible particles called atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
